
The available immune checkpoint inhibitors are usually monoclonal antibodies against these two molecules to restore the normal anti-tumor effect of T cells by blocking the interaction between these two molecules and their receptors or ligands.
8 There has been particular focus on two immune checkpoint molecules, cytotoxic T lymphocyte antigen-4 (CTLA-4) and programmed cell death protein-1 (PD-1), which have been dedicated to have potent immune suppression effects through their function as negative regulators in the process of T cell activation. 7 As a key node in the process of tumorigenesis and development, immune checkpoints such as PD-1/PD-L1, CTLA-4, TIM-3, LAG-3 and others provide us with a new therapeutic target. We call the molecules with negative immune regulation immune checkpoints. 6 Recently, it has been found that some molecules that can inhibit the function of immune cells was used by tumor cells to evade immune attack. 5 For most patients with NSCLC that cannot be treated with targeted drugs, platinum-based chemotherapy has been used as a standard treatment for a long time, and the median survival time for these patients is about one year. 4 But less than half of NSCLC patients have mutations in related tumor-driving genes including EGFR, BRAF, ERBB2 and rearrangement of ALK or ROS1, which can be blocked or inhibited by targeted medicine.
KEYNOTE 189 DRIVER
3 Targeted therapy seems to be the best option for these patients with inoperable advanced lung cancer that carry some driver gene mutation. Thirty percent of patients with locally advanced disease and 50% of patients with metastatic disease are unable to completely clear the lesions via surgery. However, only about 20% of patients with localdisease (stage I and II) have access to a radical resection. 2 Traditional lung cancer treatment methods includes surgery, radiotherapy, chemotherapy and targeted therapy. 1 Pathologically, lung cancer can be divided into two categories: small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC), the latter accounting for more than 85% of the total lung cancer. 2,093,876 new cases of lung cancer were diagnosed and 1,761,007 patients died of lung cancer in 2018, accounting for 11.6% of the total number of diagnosed cancers and 18.4% of the total number of cancer deaths. Lung cancer is the malignant tumor with the highest incidence and mortality all over the world.
Keywords: pembrolizumab, PD-1/PD-L1, KEYNOTE studies, first-line treatment, non-small cell lung cancer, NSCLC We hope pembrolizumab could become a better choice for more clinicians and benefit more patients with advanced NSCLC.
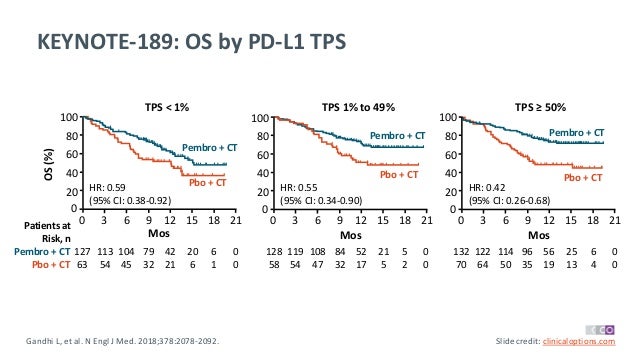
KEYNOTE 189 TRIAL
In this review, we systematically integrate the latest series of clinical trial results, pharmacological mechanisms, adverse events (AEs) and predictive biomarkers in the first-line treatment of NSCLC. Among them, pembrolizumab is currently the only PD-1 inhibitor approved for first-line treatment of NSCLC, whether it is monotherapy or combination therapy, for creditable performance in KEYNOTE studies. The discovery of immune checkpints that mediate the immune escape of tumor cells have been promoting a series of immune checkpoint inhibitors to be used in cancer treatment and achieved great results. In the past few decades, the only treatment options available for advanced NSCLC patients have been targeted therapy or chemotherapy, but these therapies are inevitably tolerated by tumors. The Affiliated Hospital of Qingdao University, Qingdao University, 16 Jiangsu Road, Qingdao 266003, People’s Republic of ChinaĪbstract: Lung cancer is currently the malignant tumor with the highest incidence and mortality in the world, while non-small cell lung cancer (NSCLC) is the most common pathological type of lung caner. *These authors contributed equally to this workĬorrespondence: Xiaochun Zhang Department of Medical Oncology Jialin Qu *, Li Wang *, Man Jiang, Deze Zhao, Yuyang Wang, Feng Zhang, Jing Li, Xiaochun Zhangĭepartment of Medical Oncology, The Affiliated Hospital of Qingdao University, Qingdao University, Qingdao 266003, People’s Republic of China

